Category Medicine and Healthcare

New Jersey Neurosurgeon Dr. Branko Skovrlj Earns Board Certification in Neurosurgery
To receive board certification in neurosurgery, candidates must complete seven years of residency training and an optional one- or two-year fellowship. Following residency, candidates complete several years practicing medicine, in which they are to log 125 cases prior to a rigorous oral and written examination.
Read Press Release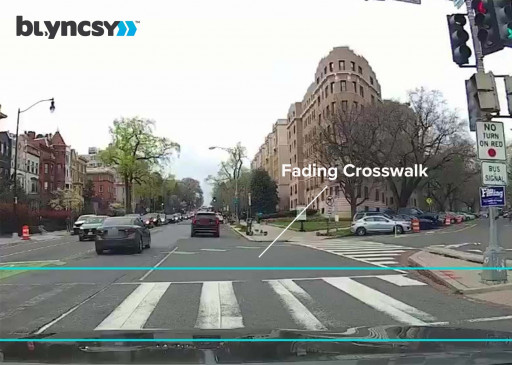
Blyncsy Joins New York City's Newlab DOT Studio to Improve Transportation
Movement data intelligence company Blyncsy joins Newlab - a community of experts and innovators applying transformative technology to solve the worldʼs biggest challenges - with the New York City Department of Transportation.
Read Press Release
National Certification Board for Behavioral Health Professionals to Administer National Certified Peer Specialist (NCPS) Professional Credentialing Program
The National Certification Board for Behavioral Health Professionals (NCBBHP) announced that going forward Mental Health America's (MHA) National Certified Peer Specialist (NCPS) program will be consolidated and managed by NCBBHP.
Read Press Release
KoreSummit Helps Companies and Entrepreneurs in the Health Industry Raise Capital Compliantly
Medtech, Biotech, and Pharma industries will gather for the Medtech A+ Team - how to do a successful RegA+ for a Medtech company in a different format, with quicker panels and more information
Read Press Release
EHIR and Magnify Ventures Join Forces to Accelerate Emerging Solutions Addressing the Needs of the Modern Family
Ten hand-selected teams will participate in their second annual Care Economy & Family Technology Academy combining a customized advisory, expert-led content, and industry exposure to emerging caregiving support solutions.
Read Press ReleaseDiabeloop - Key Player in Therapeutic AI Applied to Insulin Delivery - Announces 70 Million Euros New Financing Round to Accelerate Its International Expansion
High-growth diabetes company Diabeloop closes its Series C financing round - led by LBO France, joined by Terumo Corporation, Innovacom and backed by Diabeloop's historical partners - to power the company's commercial roll-out and global expansion.
Read Press Release
Black Book™ Announces 2022 Top Client-Rated Financial Solutions Delivering Digital Transformation and Managing Liquidity
Over 900 hospitals and inpatient organizations and 4,000 physician practices were represented in the sweeping Q1-Q2 2022 series of financial management IT user polls, as daunting challenges are forcing providers to advance their technologies to maintain solvency.
Read Press Release
Netsmart Rated the No. 1 Solutions Vendor for 8th Year, Black Book Home Health and Post-Acute Technology Users' Survey
Black Book Research estimates the North American market for post-acute software and IT services market in Q2 2022 has reached $3.0 Billion and is anticipated to grow at a CAGR of 17% during 2022-2032.
Read Press ReleaseRecardio and FDA Reach Agreement on Pivotal Phase 3 Trial Design
Recardio Inc., a late stage clinical-stage life science company developing regenerative therapies for cardiovascular diseases, announced that the FDA concurs with Recardio's pivotal Heal-MI Phase 3 trial design with Dutogliptin in Acute Myocardial Infarction.
Read Press Release
Press Conference June 2 After Historic Sentencing Hearing in Corey J. Iodice Case
● Flagman Founder Cindy Iodice Holds Press Conference June 2, Scheduled After Historic Sentencing in Slow Down Move Over (SDMO) Case ● U.S. Senator Richard Blumenthal Joins to Discuss Congressional Efforts to Promote Road Safety
Read Press Release