Category Medical Research

Concept Medical Inc. Granted 2nd 'Breakthrough Device Designation' From the FDA for Its MagicTouch PTA Sirolimus Coated Balloon, for the Treatment of Peripheral Artery Disease (PAD) in Below-the-Knee (BTK) Indication
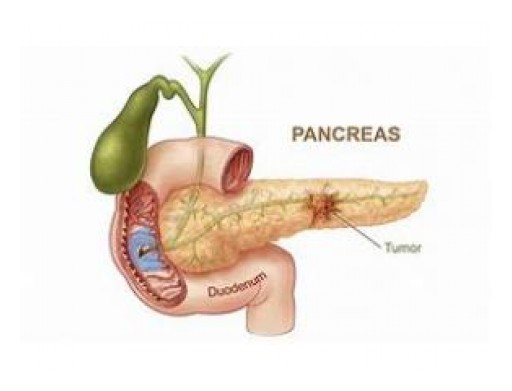
Concept Medical Inc. (CMI) has been granted "Breakthrough Device Designation" from the U.S. Food and Drug Administration (FDA) for MagicTouch PTA (Percutaneous Transluminal Angioplasty), its Sirolimus drug-coated balloon (DCB) catheter, for the treatment of Peripheral Artery Disease (PAD) in Below-the-Knee (BTK). The first 'Breakthrough Device Designation' received by Concept Medical Inc. was on 30th April 2019 for its MagicTouch SCB, for the treatment of Coronary Artery Disease (CAD) in patients with in-stent restenosis.
Read Press Release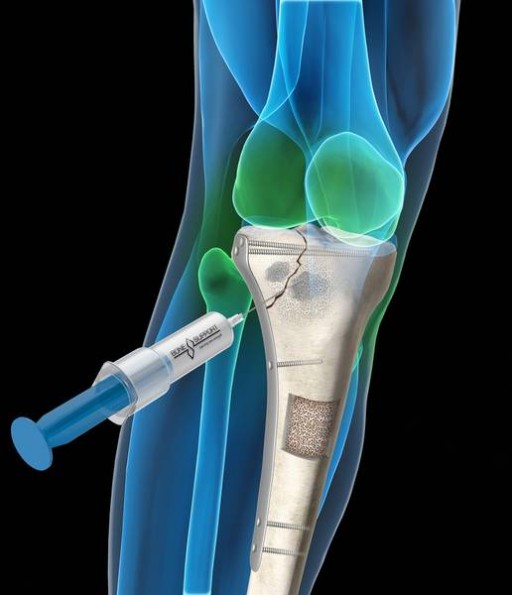
Injectable Bone Graft Market Growth 2019 - 2025: QY Research
This report studies the global market size of Injectable Bone Graft, especially focuses on the key regions like United States, European Union, China, and other regions (Japan, Korea, India and Southeast Asia).
This study presents the Injectable Bone Graft production, revenue, market share and growth rate for each key company, and also covers the breakdown data (production, consumption, revenue and market share) by regions, type and applications. history breakdown data from 2014 to 2019, and forecast to 2025.

Parnell Pharmaceuticals Holdings Ltd Announces Business Results for the Three Months Ended 30 September 2018
Parnell delivers 39% revenue growth and a $5.9 million year-to-date improvement in EBITDA as at the third quarter of 2018 compared to the same period in 2017, consolidates the return to profitability, and maintains full-year 2018 guidance at a 35 - 40% increase over 2017 revenue to $26 - $27 million and an EBITDA range of $5.5 - $6.5 million.
Read Press Release
Two Young Women Lung Cancer Investigators Receive $200k Research Grants From Lung Cancer Foundation of America and the International Association for the Study of Lung Cancer
Researchers will use IASLC/LCFA funds to create new lung cancer treatments for never-smokers and patients whose cancer has spread
Read Press Release
International Symposium Convenes to Address Ehlers-Danlos Syndromes and Hypermobility Spectrum Disorders
With generous support for research funding, The Ehlers-Danlos Society sponsors International Symposium in Ghent, Belgium, to accelerate collaborative global research into EDS, HSD, their related symptoms and associated conditions.